
Daniel Schwarz, osapiens Expert | 4. May 2026 | Lesezeit 15 min.
Is your CMMS actually ready for a regulatory audit? Incomplete records, missing electronic signatures, and unvalidated systems put your entire operation at risk. Find out how the right CMMS helps pharmaceutical industries stay inspection-ready without the administrative burden.




ENTERPRISE-READY
SAP Certification
ISO 27001
EU Hosting
Entra ID & Okta
Multi-Entity
Table of Contents
- Key Facts
- Benefits of CMMS for Pharma Industries
- What Makes a CMMS Compliant for Pharma
- Audit Trails and Data Integrity in Pharma CMMS
- What CSV Means for Your CMMS
- How to Validate a CMMS for Pharmaceutical Manufacturing
- How System Updates Are Handled: Re-Validation in a Pharma CMMS
- 5 CMMS Solutions for Pharmaceutical Manufacturing Compared
- Audit-Ready Maintenance with the osapiens HUB
- Conclusion: The Best CMMS for Pharmaceuticals
- FAQ
What does it take to pass an FDA inspection with confidence? The answer starts long before the inspector arrives, with validated systems, tamper-proof audit trails, and maintenance records that meet 21 CFR Part 11 and ALCOA+ standards. Generic CMMS platforms rarely deliver this out of the box. Life sciences manufacturers need purpose-built solutions that embed GxP compliance and Computer System Validation (CSV) directly into daily maintenance workflows. This article breaks down what those requirements mean in practice and what a pharma-ready CMMS needs to deliver.
Key Facts
- Regulatory baseline: A pharma CMMS must deliver 21 CFR Part 11 compliant audit trails, electronic signatures, and role-based access control to satisfy inspection requirements.
- Paper and Excel fail GxP: Spreadsheet and paper-based maintenance records cannot guarantee attributability, immutability, or instant retrievability, making them structurally incompatible with GxP data integrity standards.
- Choosing the right CMMS matters: Not every maintenance platform is built for pharmaceutical manufacturing. The right solution reduces audit risk, closes documentation gaps, and gives regulated industries the tools to stay inspection-ready without added administrative burden.
- osapiens HUB for Maintenance: Purpose-built for regulated industries with electronic records designed to meet 21 CFR Part 11 requirements, certified SAP ECC and S/4HANA connectors, and embedded GxP validation support from pharmaceutical experts.
Benefits of CMMS for Pharma Industries
Pharmaceutical manufacturing operates under some of the most demanding regulatory conditions of any industry. A purpose-built CMMS directly addresses the compliance and operational challenges that generic tools cannot.
- Inspection readiness at all times: Automated audit trails and tamper-proof records mean your team is never scrambling to compile documentation before an FDA or EMA inspection.
- Fewer human errors in documentation: Mandatory field configuration and digital checklists prevent incomplete work orders from being closed, eliminating the most common source of inspection findings.
- Faster response to equipment deviations: When a GMP-critical asset fails or falls out of qualification, a CMMS with full equipment history allows root cause analysis in minutes rather than days.
- Preventive maintenance on schedule: Automated reminders and scheduling ensure that no statutory inspection deadline or manufacturer-recommended service interval is missed and that every completed task is documented with a timestamp.
- Reduced validation effort over time: A cloud-based CMMS with structured change control support makes re-validation after system updates manageable, rather than a resource-intensive project that delays upgrades.
- Technician adoption that actually sticks: A mobile-first interface means technicians document work directly on the production floor, rather than returning to a desktop after the fact – which is where records become incomplete or inaccurate.
Built for pharma. Ready for any inspection
See how the osapiens HUB for Maintenance helps pharmaceutical teams stay inspection-ready – with built-in audit trails, electronic signatures, and GxP-compliant workflows out of the box.
What Makes a CMMS Compliant for Pharma
Compliance is a two-way street. Your CMMS vendor provides the technical foundation, but your organization still needs to do its part: completing IQ/OQ/PQ qualification, writing clear SOPs for system administration, and reviewing audit trails on a regular schedule. Without that internal layer, even the most capable system will struggle under FDA scrutiny.
On the technical side, a pharma-compliant CMMS needs to meet several key requirements:
- 21 CFR Part 11: Tamper-proof audit trails, electronic signatures legally equivalent to handwritten ones, unique user IDs, and documented system validation to ensure accuracy and reliability.
- 21 CFR 211.67/211.68: Complete records for equipment cleaning and maintenance, including written procedures, cleaning schedules, and records for every piece of equipment used in drug manufacturing.
- EU GMP Annex 11: Full lifecycle management of computerized systems, including cybersecurity as a core GMP requirement under the upcoming revision.
- ALCOA+ principles: All maintenance data must be Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, and Available.
- Mandatory field configuration: Required fields must stop technicians from closing work orders before all information is filled in. Incomplete records that are marked as complete remain the most common reason for FDA inspection failures.
This is why generic CMMS platforms that allow record deletion or editing without a traceable log represent a serious compliance risk for any pharmaceutical manufacturing operation.
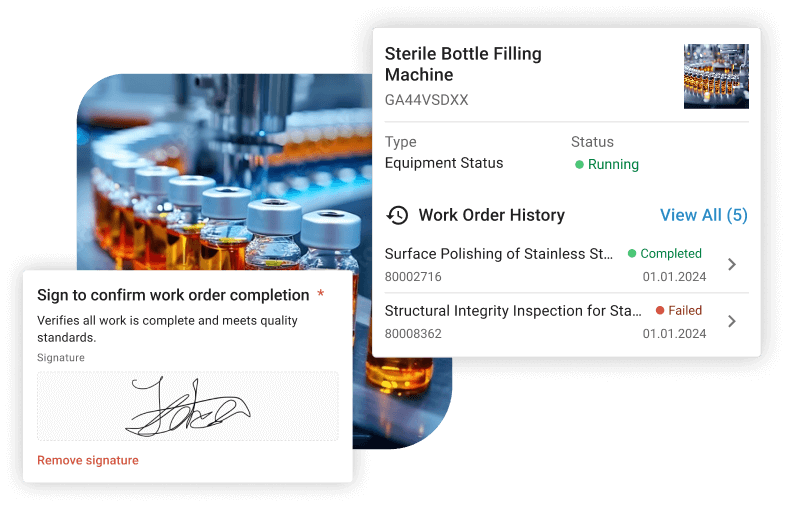
Companies like Boehringer Ingelheim and Bayer rely on osapiens to keep their maintenance operations audit-ready without sacrificing technician productivity.
What System Environments Does a Pharma CMMS Require?
Running a validated CMMS in a pharmaceutical environment means more than having the right software. It means operating it in a controlled, structured system landscape. Most regulated organizations work with three environments, each with a distinct purpose.
- The development and test environment is where configuration changes, new features, and system adjustments are tested before they move any further. Nothing here affects validated functionality or live data.
- The validation environment is where formal qualification activities take place. IQ, OQ, and PQ protocols are executed here, and all test evidence is captured in this environment before any change is cleared for production. Keeping this environment separate from both development and production is a regulatory expectation, not just a best practice.
- The production environment is the live, validated system where all GMP-relevant maintenance activities take place. Only changes that have successfully passed through the validation environment, with documented evidence and sign-off, are promoted to production.
This three-environment structure ensures that no testing or qualification activity ever interferes with validated production data, and that your validated state remains intact at all times.
Audit Trails and Data Integrity in Pharma CMMS
An audit trail in maintenance automatically and chronologically documents every action related to maintenance, repair, and inspection. It captures who performed an action, when it took place, and what changes were made, including whether those changes affected the qualification or validation status of the asset.
Organizations with complete documentation across all six areas pass audits with confidence. Those with gaps risk findings, costly rework, or in the worst case, production shutdowns.
An audit trail in a pharmaceutical CMMS is not a feature you simply switch on and forget. Both 21 CFR Part 11 and EU GMP Annex 11 treat audit trails as a regulatory expectation that demands routine review. Every modification to maintenance data must be captured automatically, and your quality team must regularly verify that these logs remain complete and unaltered.
| Audit Trail Element | What It Records |
|---|---|
| Original value | The data before any change |
| New value | The data after the change |
| Date and time | Computer-generated timestamp |
| User ID | Unique identification of the person making the change |
| Meaning statement | Reason for the change |
Routine audit trail review needs to be documented as part of your quality system, not treated as an optional activity that only happens before an inspection.
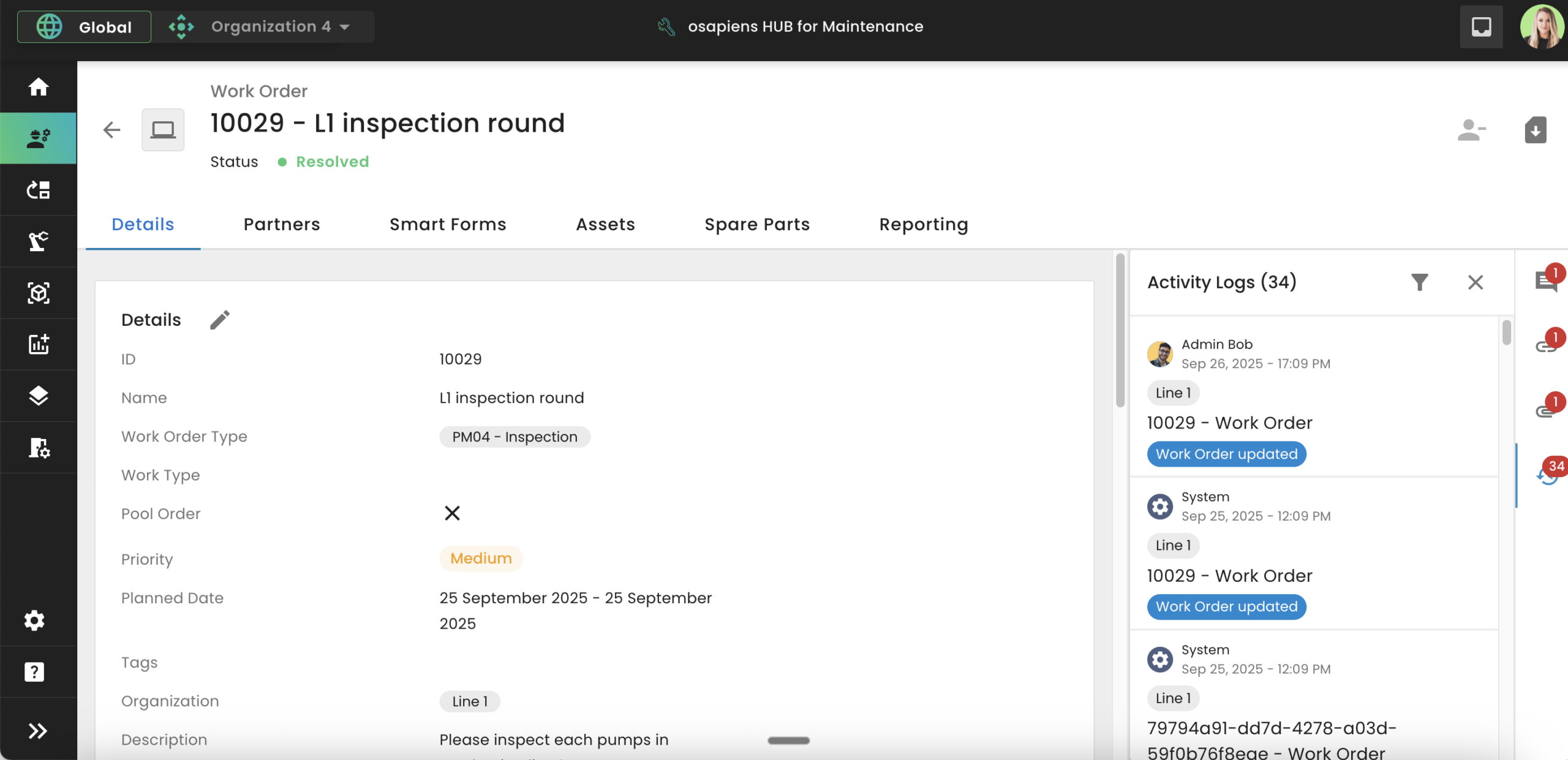
A digital audit trail in a CMMS like the osapiens HUB for Maintenance addresses the root cause:
✓ Automation instead of manual documentation
✓ Every action is logged automatically
✓ Changes are immutably traceable
✓ The complete asset history is available at any time
Audits become a routine task rather than a source of stress, because all evidence is already captured in the system.
What CSV Means for Your CMMS
Computer System Validation (CSV) ensures that a software system does what it is supposed to do reliably, consistently, and in a documented way. In pharmaceutical manufacturing, this requirement applies directly to your CMMS. Any new implementation must be validated before go-live, and any significant change to the system triggers a re-validation.
Three regulatory frameworks define what CSV must achieve in practice:
| Framework | Scope | Key Requirements for CMMS | Relevance |
|---|---|---|---|
| GAMP 5 | International | Risk-based validation; CMMS = Category 4 (configurable software); requires IQ, OQ, PQ documentation and configuration evidence | De-facto global standard for structuring the validation process |
| FDA 21 CFR Part 11 | USA / FDA-regulated markets | Tamper-proof audit trails, strict access controls, electronic signatures legally equivalent to handwritten ones | Mandatory for any facility subject to FDA inspection |
| EU GMP Annex 11 | European Union | Supplier qualification, periodic system reviews, ALCOA+ data integrity, cybersecurity controls; revised draft expands scope significantly | Mandatory for all pharma production and distribution within the EU |
In practice, a CSV-compliant CMMS must deliver on five core requirements:
- A complete audit trail across all data entries and changes
- Electronic signatures for approvals that meet FDA 21 CFR Part 11
- Role-based access controls to prevent unauthorized modifications
- Tamper-proof records of all maintenance activities
- Documented change control processes for every software update
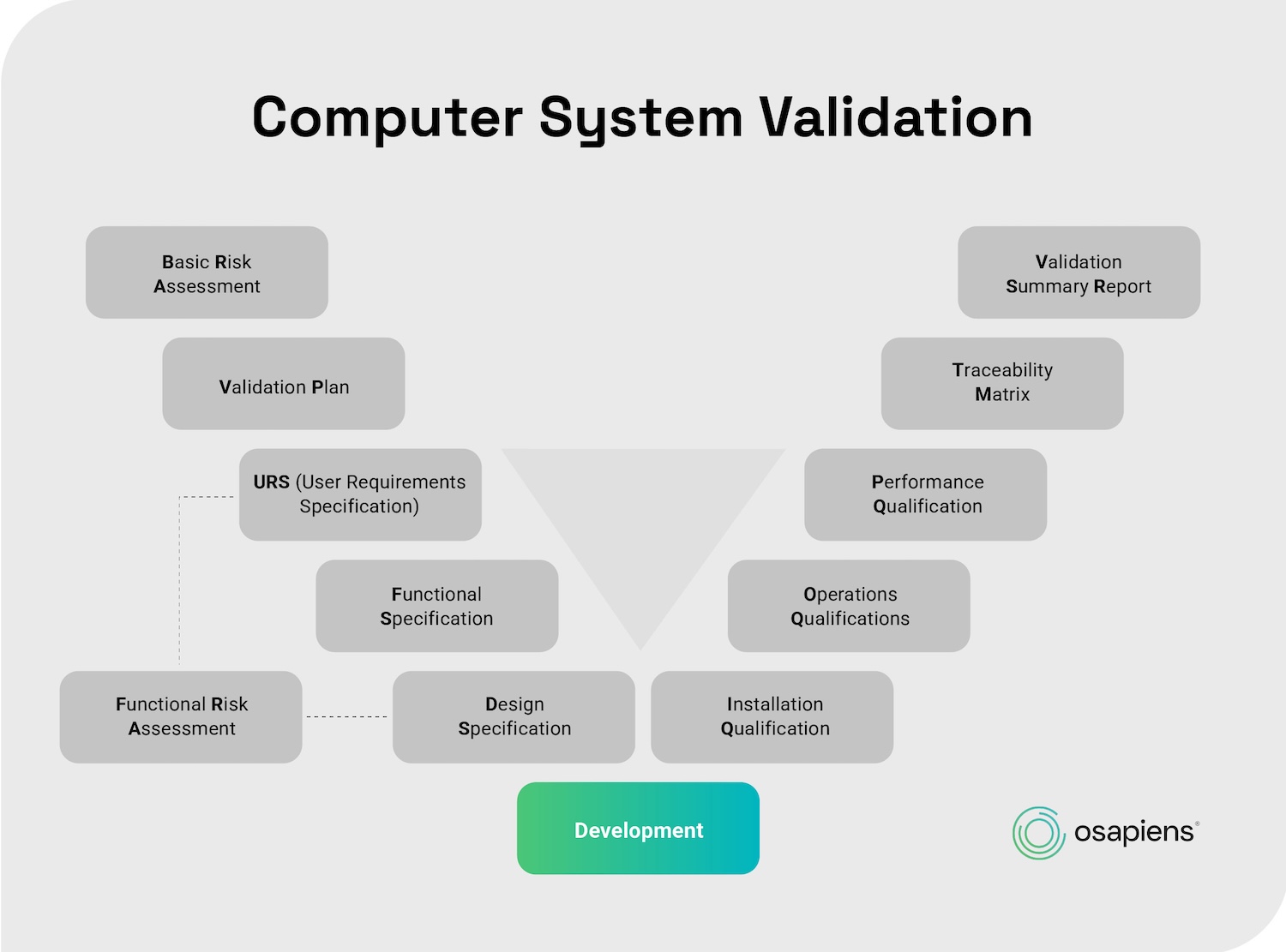
The V-model provides the standard framework for structuring CMMS validation in pharmaceutical environments.
How to Validate a CMMS for Pharmaceutical Manufacturing
Validating a CMMS sounds complex, and it can be if you go in without a clear process. In practice, it follows four phases that take you from initial planning all the way through go-live and beyond.
1. Define your User Requirements Specification (URS)
Start by writing down exactly what the system needs to do. Your quality and maintenance teams should align on both compliance requirements such as audit trails, electronic signatures, and access controls, as well as day-to-day operational needs like work order management and SAP integration. Everything you test later will be measured against this document.
2. Installation Qualification (IQ)
IQ confirms the system was set up correctly. For a cloud-based CMMS, this means verifying the software version, configuration settings, and the infrastructure documentation your vendor provides.
3. Operational Qualification (OQ)
OQ checks that the system does what it is supposed to do. You test every compliance-relevant function including audit trail generation, electronic signatures, access controls, and mandatory fields against your URS requirements.
4. Performance Qualification (PQ)
PQ is where you run the system through real workflows: a full preventive maintenance cycle, a corrective work order with deviation documentation. The goal is to confirm that records are complete, attributable, and retrievable under actual working conditions.
Validating a CMMS does not have to be a project your team figures out alone. The osapiens HUB for Maintenance comes with pharmaceutical experts who have been through this process many times and know where teams typically get stuck.
Traceability Matrix: The Thread That Connects Your Entire Validation
Completing IQ, OQ, and PQ is not enough on its own. Auditors do not just want to see that tests were run. They want to see that every requirement in your URS was actually tested and that the evidence is traceable. That is exactly what a traceability matrix delivers. It maps each URS requirement to the corresponding test cases across IQ, OQ, and PQ, and documents whether each test passed, failed, or raised a deviation. The result is a single document that gives auditors complete visibility into your validation coverage at a glance.
The most common mistake teams make is building the traceability matrix after the fact, once all testing is done. By that point, gaps are harder to close and deviations are harder to justify. Building it in parallel with your test protocols keeps your validation airtight from the start.
How System Updates Are Handled: Re-Validation in a Pharma CMMS
A validated CMMS does not stay validated automatically. Every change to the system, whether a new software version, a configuration adjustment, or an added module, has the potential to affect validated functionality.
In practice, every change follows four steps:
- Change Request: Any planned modification to the system is formally documented before it is implemented. This includes a description of the change, the reason for it, and the systems or processes it affects.
- Impact and Risk Assessment: The change is evaluated against the validated state of the system. The key question is whether the change touches any functionality that was previously validated. Low-risk changes, such as minor UI adjustments, may require only a brief documented assessment. Changes that affect audit trails, electronic signatures, or access controls trigger a full re-qualification.
- Approval: Before implementation, the change must be reviewed and approved by the relevant stakeholders, typically quality assurance and the system owner. No change goes live without documented sign-off.
- Documentation and Re-Validation: Once approved and implemented, the change is documented in the system’s validation lifecycle file. If re-qualification is required, the relevant IQ, OQ, or PQ protocols are re-executed and the traceability matrix is updated accordingly.
5 CMMS Solutions for Pharmaceutical Manufacturing Compared
Choosing the right CMMS for a regulated environment goes well beyond feature lists. The platforms below are among the most commonly evaluated options in pharmaceutical manufacturing, each with different strengths depending on your compliance requirements, existing infrastructure, and operational priorities.
- osapiens HUB for Maintenance is a European CMMS purpose-built for regulated industries. Its core value proposition combines deep, SAP-certified integration with native GxP compliance. SAP remains the system of record for financial and planning data, while the osapiens HUB provides an intuitive technician layer that eliminates the shadow systems and paper workarounds that create audit risk. The platform emphasizes data integrity and inspection readiness, with built-in audit trails, electronic signatures, and ALCOA+ compliant documentation aligned with 21 CFR Part 11 and EU GMP Annex 11 requirements.
- IBM Maximo is an established enterprise asset management platform with a long track record in heavily regulated industries. It offers native 21 CFR Part 11 capabilities and full audit trails, and is well-suited for large-scale, multi-site deployments. Trade-offs include complex implementation requiring significant consulting investment, a steep learning curve for shop-floor technicians, and SAP integration that requires third-party middleware.
- SAP Plant Maintenance (SAP PM) is the standard maintenance module within the SAP ERP ecosystem and remains widely used across large pharmaceutical enterprises. Its primary strength lies in its deep integration with finance, procurement, and supply chain processes, ensuring end-to-end data consistency and compliance at scale. SAP PM supports audit trails and can be configured to meet GxP and 21 CFR Part 11 requirements, but achieving this typically requires significant customization and validation effort. While powerful from a system-of-record perspective, its user experience is often not optimized for shop-floor technicians, which can lead to the adoption of parallel tools or manual workarounds without an additional execution layer.
- eMaint offers a dedicated life sciences CMMS with a notable CSV validation service to support the computer systems validation process. Its strengths include configurable reporting, calibration tracking, and preventive maintenance automation, all of which support audit readiness and regulatory inspections. SAP integration typically requires third-party connectors, and while the mobile experience is functional, it is not built with a mobile-first philosophy. This can impact technician adoption on the shop floor.
- Limble CMMS is a US-based maintenance platform with a dedicated version for regulated industries. It supports 21 CFR Part 11 compliance with audit trails, electronic signatures, and access controls, and comes with pre-built validation documentation including IQ/OQ/PQ protocols and a traceability matrix. Its interface is intuitive and technician-friendly, which supports adoption on the shop floor. For European pharmaceutical operations, however, explicit EU GMP Annex 11 alignment is less clearly documented, and SAP integration requires third-party connectors.
Audit-Ready Maintenance with the osapiens HUB
Pharmaceutical companies need a CMMS that meets the strict requirements of regulated industries like pharma and life sciences. The osapiens HUB for Maintenance was built as a SaaS solution for exactly this purpose, helping pharma teams unite compliance and operational efficiency in a single platform. The platform combines native GxP compliance, SAP-certified integration, and a mobile-first experience in a single platform, without having to patch together separate tools.
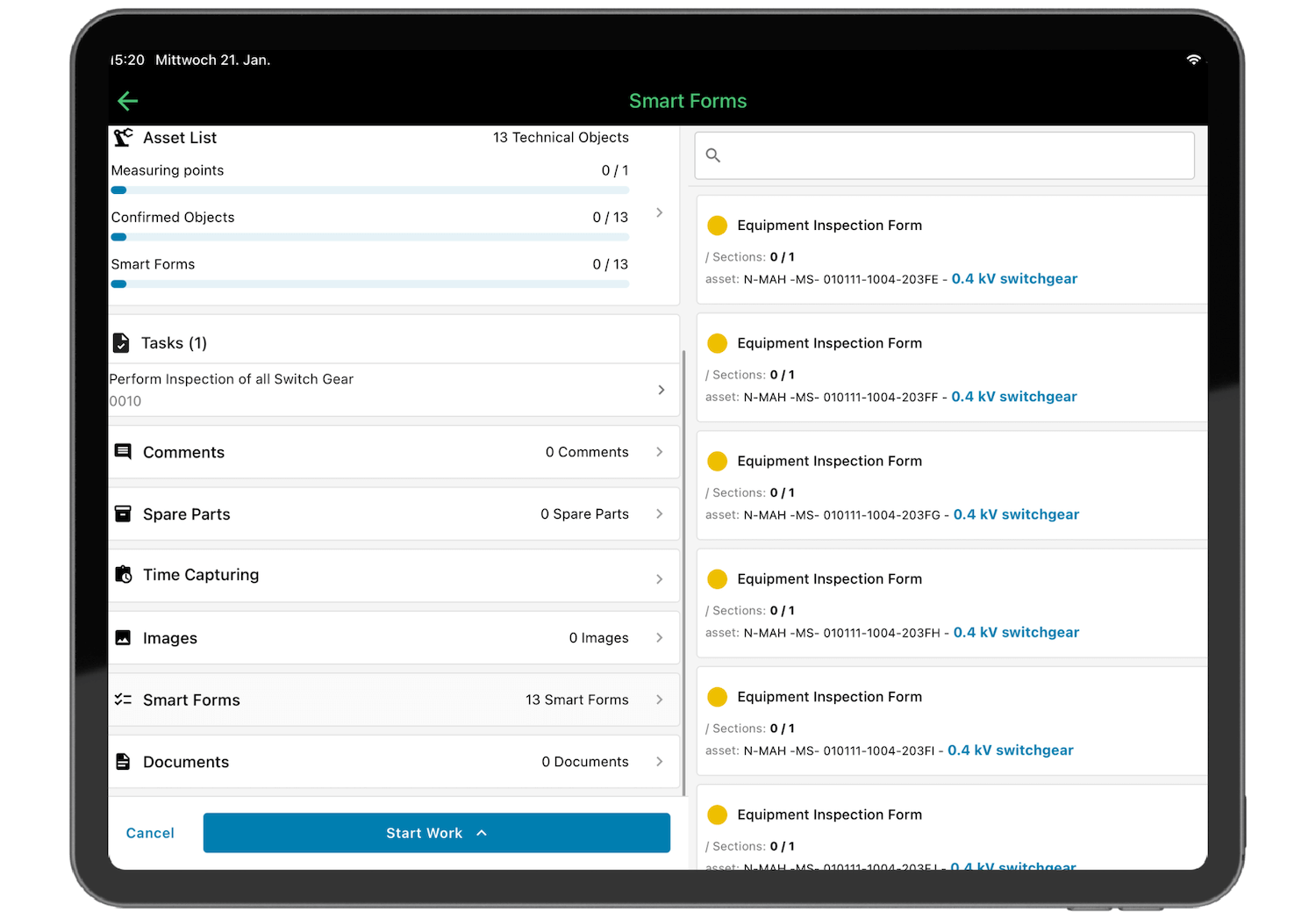
The core features with direct relevance to pharma compliance include:
- Immutable audit trails covering all activities and data changes for complete traceability
- Electronic signatures for maintenance approvals in line with 21 CFR Part 11
- Role-based access controls to protect sensitive GxP data from unauthorized changes
- Digital checklists and structured documentation for tamper-proof maintenance records
- SAP-certified integration for consistent data flow between your CMMS and ERP system
- Power BI connectivity to turn maintenance data into enterprise-wide reporting and precise KPI tracking
- Mobile app that lets technicians complete documentation directly on the production floor
Leading pharmaceutical manufacturers like Boehringer Ingelheim and Bayer already trust osapiens to keep their maintenance operations compliant by default, so their teams can focus on the work that matters.
See pharma-compliant workflows in action
See how the osapiens HUB handles Part 11 audit trails, electronic signatures, and preventive maintenance scheduling in a live walkthrough tailored to your regulatory environment.
Conclusion: The Best CMMS for Pharmaceuticals
The right CMMS for pharmaceuticals is not a maintenance tool with compliance features bolted on. It is a compliance platform that enables efficient maintenance. Every workflow, every audit trail entry, and every electronic signature must be designed with regulatory scrutiny in mind from the start. The best choice depends on three factors: compliance depth, SAP integration needs, and technician usability on the shop floor.
For SAP-centric pharma organizations that require native 21 CFR Part 11 compliance, mobile-first execution, and dedicated pharmaceutical validation support, the osapiens HUB for Maintenance delivers the strongest combination of these capabilities. Digital maintenance documentation is no longer a competitive advantage. It is rapidly becoming the regulatory baseline that every pharmaceutical plant must meet.
FAQ
What system environments are required for a pharma CMMS?
A pharma CMMS operates across three separate environments: a development and test environment for configuration and feature testing, a validation environment for formal IQ, OQ, and PQ activities, and a production environment for all live GMP operations. This separation ensures that no testing or qualification activity ever interferes with validated production data.
Can a cloud-based CMMS meet GxP compliance?
Yes. A cloud-based CMMS can be GxP-compliant when the vendor provides supplier qualification documentation and the organization completes its own CSV or CSA validation process. Cloud platforms also benefit from the FDA’s CSA guidance, which simplifies revalidation cycles compared to on-premise systems.
How long does CMMS implementation take in pharma?
Software deployment typically takes weeks. However, the validation effort, including IQ/OQ/PQ qualification, adds weeks to months depending on system complexity and organizational readiness. The osapiens HUB for Maintenance offers dedicated pharmaceutical expert guidance that supports both deployment and the validation process, helping teams move through each phase efficiently.
What is the fastest way to validate a CMMS in pharma?
Start with a clearly written URS that captures both compliance and operational requirements. From there, IQ, OQ, and PQ follow a structured sequence. Working with a vendor that provides pharmaceutical expert guidance significantly reduces the time and internal effort required, particularly for teams validating a CMMS for the first time.
Start using osapiens for free
Improve your uptime, extend the life of your equipment, and simplify work order management.
ENTERPRISE CAPABILITIES
- SAP ECC & S/4HANA Certified
- Infor & MS Dynamics
- Microsoft Entra ID / Okta
- ISO 27001 & 9001
- EU Cloud · Data Sovereignty
- Development, Test, and Production Environments
- Multi-Entity & Multi-Site
- Power BI Integration
- Audit Trail & Compliance
Related content
Powder Coating Equipment Maintenance Checklist – Tasks, Safety & Digital Execution

Circuit Breaker Maintenance Checklist – Tasks, Tips & Template
Scheduling Software for Field Service: How to Optimize Dispatching

How to Ensure a Successful CMMS Implementation

Asphalt Paver Maintenance Checklist – Tasks, Systems & Digital Execution

Fleet Maintenance Checklist – Tasks, Tips & Template

CMMS Migration Made Simple: How to Switch Maintenance Software Without Data Loss

Work Order Software: How Modern Solutions Transform Maintenance Operations

Bioreactor Maintenance Checklist – Tasks, Compliance & Digital Execution

Blister Packaging Machine Maintenance Checklist – Tasks, Components & GMP Compliance

Chilled Water Pump Maintenance Checklist – Tasks, Tips & Template












