Blog
Welcome to our company blog! Here, you’ll find valuable insights, practical tips and in-depth knowledge on supply chain transparency, operational efficiency, smart maintenance, and regulatory compliance. Stay informed about key developments and discover new ideas to drive sustainable growth across your business.
Filtering by:

Beyond Compliance: Why Sustainability is Driving Innovation Across Europe
In today’s fast-changing business environment, sustainability has evolved from a compliance requirement into a powerful driver of innovation. Forward-thinking companies across Europe are leading the way: redefining their business models, …
June 23, 2025
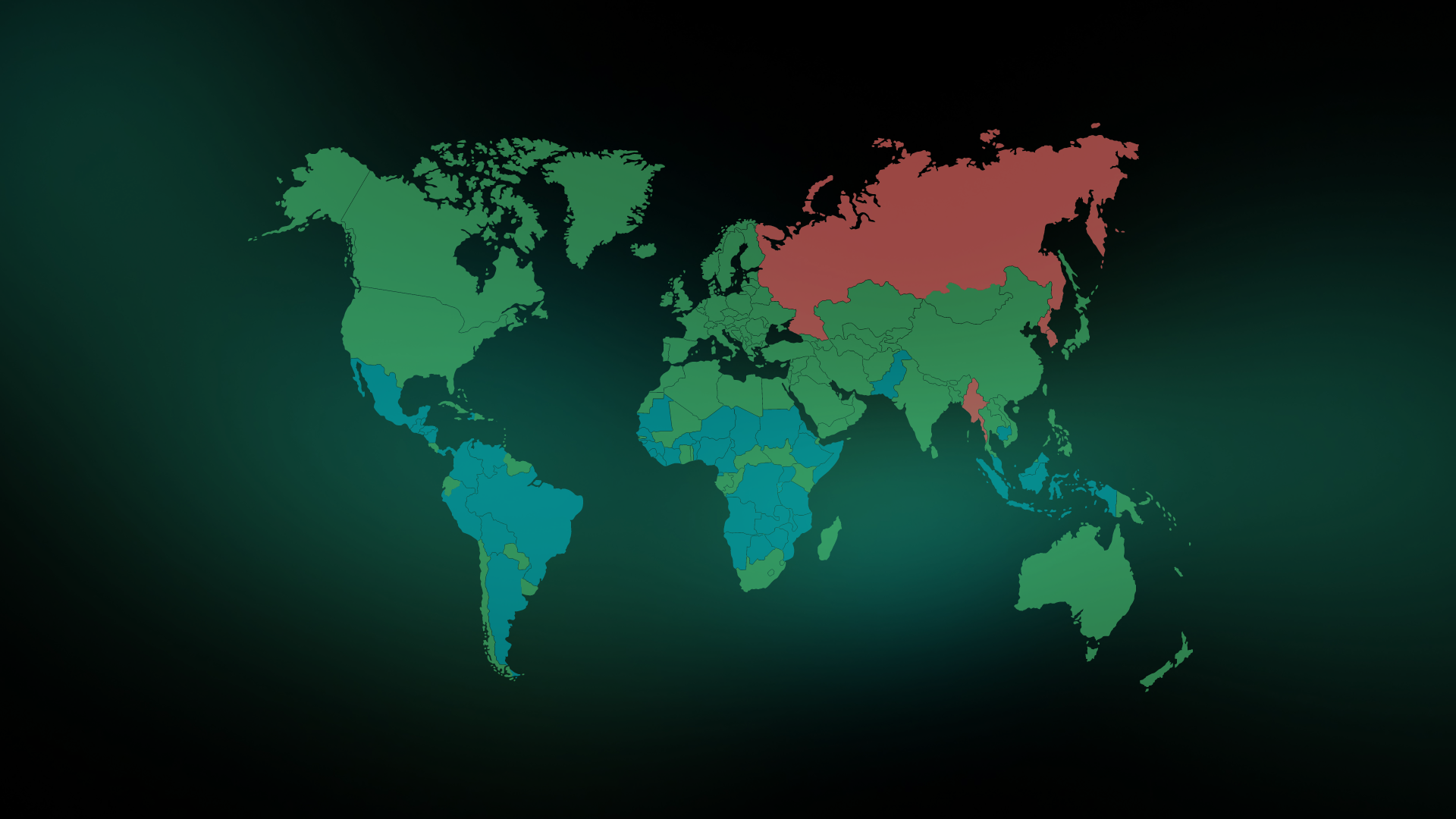
Navigating the EU’s EUDR Benchmarking System: What It Means for Your Supply Chain
As the EU Deforestation Regulation (EUDR) moves closer to full implementation, businesses across the globe are preparing to meet its stringent due diligence requirements. A key component of this regulation …
June 20, 2025
Avoid Costly Mistakes: Critical Pitfalls in EUDAMED Registration
EUDAMED compliance does more than satisfy regulators – it secures your access to the European market. Even a single invalid data point or misclassified identifier can delay or block product …
May 28, 2025

Traceability Made Easy: Ensuring Your Products Meet Current EUDR (FAQ Version 4) Standards
On April 16, 2025, the EU released FAQ Version 4 for the EU Deforestation Regulation (EUDR). This document clearly establishes the rules for placing certain commodities and products on the …
May 22, 2025

Omnibus Package and “Stop-the-Clock”: What Changes, What Doesn’t — and Why Your Sustainability Efforts Still Count
Many companies across Europe have already taken significant steps to prepare for the CSRD, CSDDD, CBAM, and EU Taxonomy. Supplier data has been gathered, double materiality assessments are underway, and …
May 21, 2025

EUDR is Here – What’s Next? Your Essential Checklist for Taking Action
EUDR will soon become a reality for thousands of companies in Europe — and for those ready to see the bigger picture, it can mark the beginning of their journey …
April 29, 2025

EUDAMED Countdown: Why Medical Device Manufacturers Must Act Now
Time is ticking. Medical device manufacturers face a hard deadline: by January 2026, the use of several core modules of EUDAMED will become mandatory. Manufacturers must act decisively in 2025 …
April 28, 2025

The Brightside of EUDR: Turning Regulation into Opportunity
The EU Deforestation Regulation (EUDR) is setting new standards for supply chain accountability — a shift that is reshaping how companies approach transparency. Companies are now required to prove that …
April 15, 2025


